Biotech & Life Sciences
-
Mabwell Receives FDA IND Clearance for Novel IBD Antibody 9MW5211
The Shanghai-based biopharmaceutical company announced this week that 9MW5211 received FDA IND clearance to begin clinical studies in inflammatory bowel disease. Simultaneously, China’s National Medical Products Administration accepted clinical trial applications for multiple indications including IBD and multiple sclerosis for review. The compound is described as the world’s first clinical-stage drug candidate for its specific
-
Biotech & Health Weekly: The Week a New Drug Class Got Approved
Thirteen years of research. One FDA approval. Arvinas crossed the finish line on May 1 with the first PROTAC therapy ever cleared by a regulator. The same day, Eli Lilly reported the most dominant quarter in GLP-1 history. A week later, Novo Nordisk answered with its own numbers. The biotech M&A machine kept running in
-
Australia Just Approved the First Drug to Prevent Hearing Loss From Childhood Chemotherapy
Cisplatin works. That is both its value and its problem. The chemotherapy drug has been a cornerstone of treatment for childhood solid tumors for decades. It is effective against a wide range of cancers including hepatoblastoma, neuroblastoma, and osteosarcoma. It is also one of the most reliably ototoxic drugs in oncology, meaning it damages the
-
Australia Just Approved the First New Therapy for Chronic GVHD
Chronic graft-versus-host disease develops when donated stem cells attack the patient’s own body. It affects 40 to 50% of stem cell transplant recipients. In most cases three or more organs are involved. For patients who have already failed two lines of treatment, the options until now have been limited and the outcomes often poor. Australia
-
The World’s First AI-Designed Drug Just Got Cleared for Direct-to-Lung Trials in IPF
Idiopathic pulmonary fibrosis kills most patients within three to five years of diagnosis. There is no cure. The two approved treatments slow the scarring but do not stop it, and both carry significant side effects that limit tolerability. Rentosertib was designed by an AI. Not assisted by an AI. Designed by one, from target identification
-
The Drug That Could Make GLP-1 Injections Optional Just Hit a Key Milestone
The GLP-1 drug market is already worth over $50 billion annually. Every major player in it requires an injection. That is the gap ASC30 is targeting. Ascletis Pharma (HKEX: 1672) announced on April 26, 2026 that it has completed enrollment in its 13-week US Phase II study of ASC30, an oral small molecule GLP-1 receptor
-
1 in 3 Americans Say AI Caught What Their Doctor Missed — Here’s What the Data Shows
One in three Americans say AI caught a health problem their doctor missed. Nine in ten of those conditions were later confirmed by a healthcare provider. Those are not small numbers. And they come from a survey of 1,000 adults conducted by Testing.com in April 2026, using the Pollfish platform with attention checks and fraud
-
OZMOSI and Planview Just Solved Pharma’s Biggest Planning Problem
Pharmaceutical R&D has a data problem that has nothing to do with a shortage of data. Drug developers sit on enormous volumes of information: clinical trial registries, regulatory filings, scientific literature, competitor disclosures, pipeline announcements. The problem is that it lives in disconnected silos, built on inconsistent taxonomies, in formats that resist machine-readable analysis. A
-
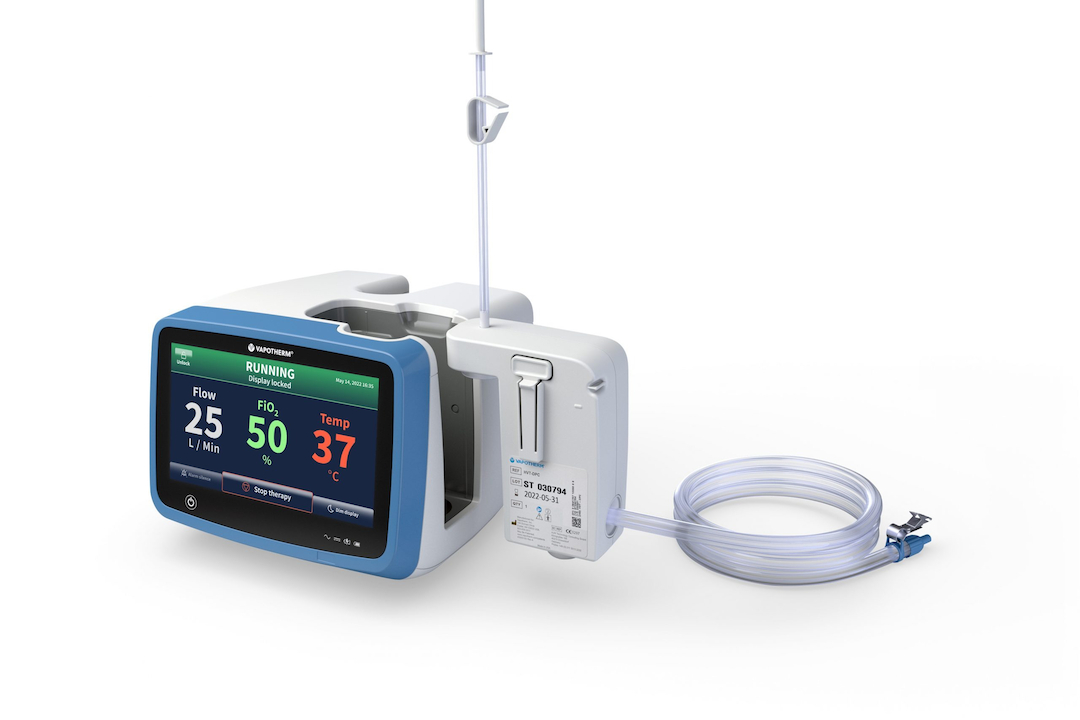
Vapotherm Just Solved a Problem Every ICU and NICU Shares
A premature infant in the NICU and a 70-year-old in the ICU need completely different respiratory support. Different flow rates, different circuit sizes, different setup requirements. Until now, that meant different disposable circuits, different inventory, different workflows for clinical staff managing both populations at once. One FDA clearance just changed that. On April 21, 2026,
-
Tumors Were Hiding Immune Cells. Atera Just Found Them.
A new instrument just removed the biggest constraint in cancer biology research. Here’s why that matters. To understand how cancer develops, you need to know three things simultaneously. Which genes are being expressed in a cell. Which specific cell is expressing them. And exactly where in the tissue that cell sits. The problem is that